Bayer and federal safety officials have pulled nearly 800,000 travel-size bottles of Afrin Original nasal spray from the market after discovering the tiny bottles are not child-resistant, which could let a young child get into the medicine and suffer serious poisoning. The voluntary recall, announced April 30, 2026, covers specific 6 mL bottles, and anyone who has an unexpired one is being urged to keep it far from kids and follow Bayer's refund and disposal steps.
What was recalled
The U.S. Consumer Product Safety Commission says the recall covers unexpired Travel Size Afrin® Original Nasal Spray 6 mL bottles, about 786,100 units in total. Affected lot numbers are 230361, 240822, 241198, 250066, 250152, 250646 and 250831. According to the agency, the travel bottles contain an imidazoline and their packaging is not child-resistant, which poses a risk of serious injury or illness if a young child swallows the contents.
Bayer's explanation and refunds
Bayer said in its press release that the 6 mL travel bottles were designed with an easy-open closure meant to help older adults and people with disabilities, but they do not include the required front-label warning "Package Not Child-Resistant." The company has notified distributors and, in coordination with the CPSC, is arranging returns of unsold product. Consumers with an unexpired bottle can request a refund online through Bayer's recall portal, which asks for a photo of the product before it is thrown away.
Where and when they were sold
The CPSC reports that the travel-size bottles were sold from September 2024 through April 2026 at convenience stores and travel hubs, including airports, typically for about $7 to $9. Regulators say there have been no injuries reported so far, but they are urging anyone with an unexpired recalled bottle to keep it out of children's reach while they begin the refund and disposal process.
Why manufacturers must use child-resistant packaging
Federal law requires "special packaging" that is child-resistant yet still reasonably easy for adults to use for certain household substances, including nasal decongestants that contain imidazoline. The standard and testing requirements are detailed in federal regulations available through Cornell Law School.
What to check and how to act
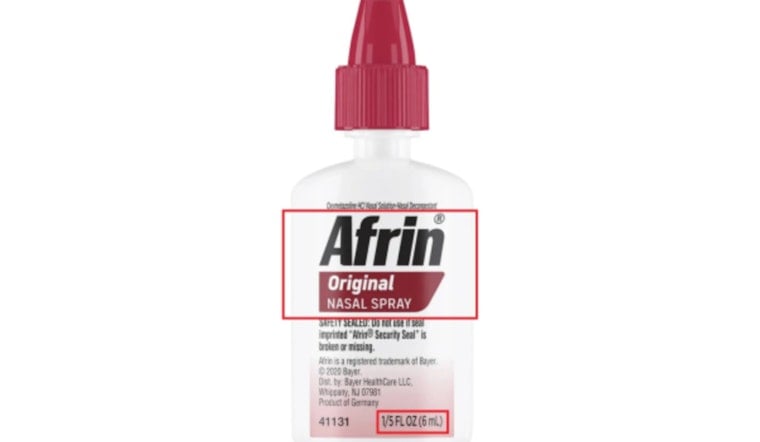
Consumers should look for small Afrin bottles labeled "AfrinAE Original Nasal Spray" with the size marking "1/5 FL OZ (6 mL)," along with a six-digit lot code and an expiration date printed in the format "YYYYMMM." If the lot number on your unexpired bottle matches one of the recalled codes, use Bayer's recall webform to request a refund and then dispose of the product as instructed, or call Bayer at 1-800-317-2165 if you have questions.









