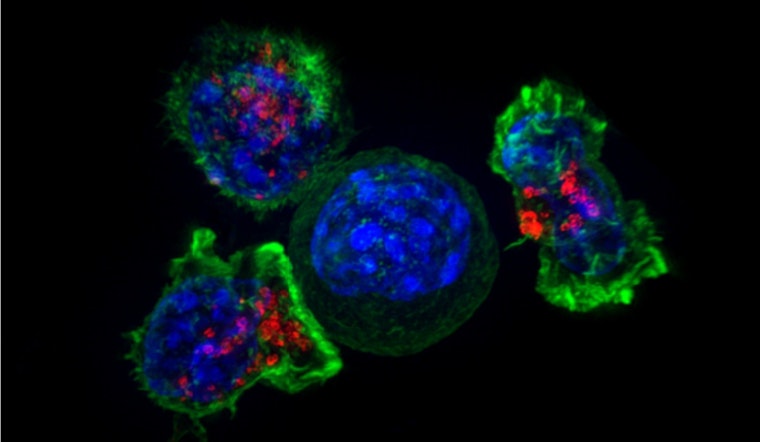
Scientists at UC San Diego, the Salk Institute, and the University of North Carolina have drawn up a detailed genetic atlas that shows how CD8 “killer” T cells decide whether the keep fighting tumors or slide into exhaustion. Using that map, the team flagged two transcription factors that, when switched off, allow exhausted T cells to regain their tumor-killing power while still preserving long-term immune memory. The work, published this week in Nature, points to fresh ways to design CAR T and adoptive cell therapies.
How the atlas maps T cell fate
The study, published in Nature today, built a multi omics atlas that integrates transcriptional and epigenetic data across nine CD8+ T cell states, according to Nature. Using a computational pipeline called Taiji together with in vivo CRISPR perturbation screens, the researchers generated transcription factor “fingerprints” for each state. That setup let them predict which regulators push cells toward protective tissue-resident memory programs and which send them into terminal exhaustion.
San Diego labs helped lead the effort
Researchers at UC San Diego, the Salk Institute, and the University of North Carolina led the project, with co-corresponding authors Wei Wang (UCSD), Susan Kaech (Salk), and H. Kay Chung (UNC), as reported by UC San Diego Today. Wang described the atlas as a step toward precisely manipulating immune cell fates, and Kaech said it lets scientists give T cells much clearer instructions, according to the university. The paper also cites multiple NIH grants that supported the research.
A pair of genetic switches
The team zeroed in on two previously unlinked transcription factors, ZSCAN20 and JDP2, whose deletion restored tumor-killing activity in exhausted CD8+ T cells without wiping out memory, as reported by the Salk Institute. In mouse models, targeted deletion of these factors reduced tumor burden and boosted responses when combined with PD 1 checkpoint blockade. Co corresponding author H. Kay Chung described the work as flipping specific genetic switches to uncouple killing function from long term protection.
What this means for CAR T and cell therapy
By separating exhaustion programs from protective ones, the study raises the prospect of engineering T cells that keep their punch without burning out, the researchers say. According to Nature, perturbing these TEX_term selective factors improved tumor control and strengthened effector function in human T cells ex vivo. The authors suggest the platform could speed up the creation of precise “TF recipes” to program cells for adoptive cell transfer and CAR T applications.
San Diego's cell therapy ecosystem
UC San Diego's role in the study is significant as the region is already expanding its cellular therapy offerings. UC San Diego Health recently rolled out personalized TIL therapy and maintains CAR T programs, highlighting local capacity to move lab discoveries into clinical trials. Local clinicians and trial networks will be central to testing whether transcription factor-based programming can be delivered safely in patients, experts say. If the “recipe” strategy holds up, it could shorten the path from genetic insight to bedside testing.
Caveats and next steps
The findings are preclinical. Most functional experiments were run in mouse models and ex vivo human cells, so safety, delivery and off target effects remain major unanswered questions, as UC San Diego Today notes. The team plans to pair advanced lab methods with AI guided computational modeling to expand and refine genetic recipes for programming T cells, the researchers say. Any future therapy will need rigorous translational work and full clinical trials before it can reach patients.
For San Diego researchers and the wider immunotherapy community, the atlas functions as a practical roadmap. It does not promise instant cures, but it hands scientists clearer levers to test first in the lab and eventually in the clinic. The coming rounds of work will show whether those levers can be turned into safe, durable treatments.









